 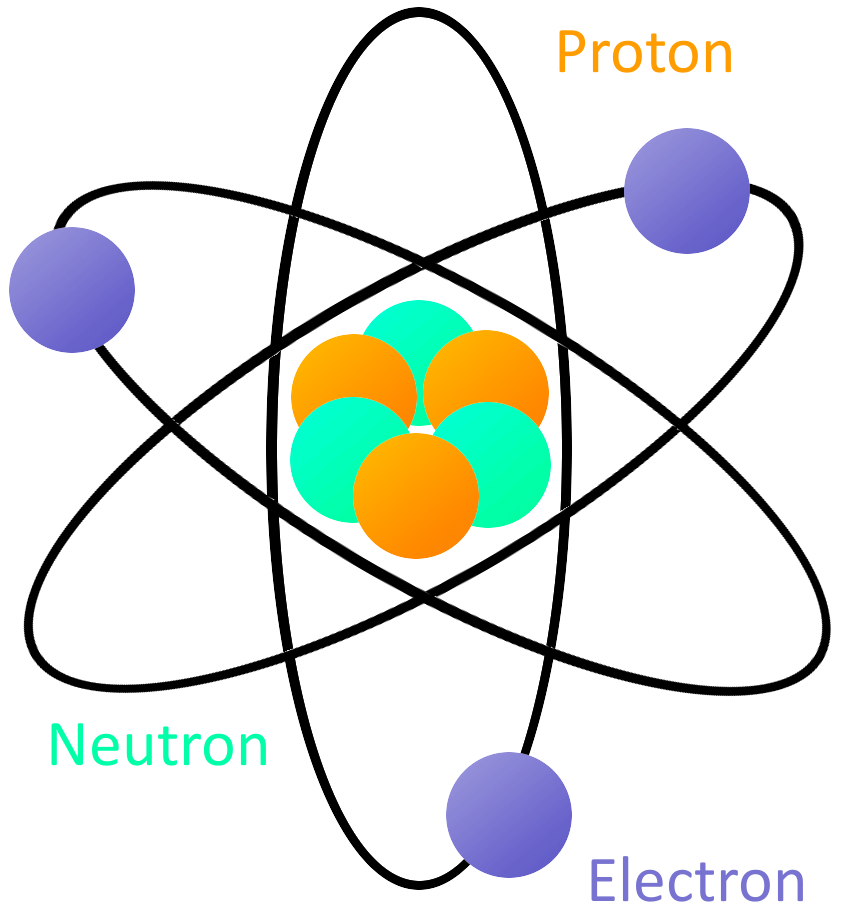
Students may be interested in the even smaller particles of quantum physics-like quarks, bosons, neutrinos, and antineutrinos. Using the dropdown, you may view a simplified model of its electron cloud or a 3D representation of the bohr model. On the left is a 3D model of the atom selected in the periodic table. They are intended to get players thinking about what they already know about elements. The electron orbital simulator (EOS) is a Unity program that serves as a visual aid for learning the structure of the atom. General questions about the properties of elements assume standard temperature and pressure (helium is liquid below -268☌ and gold is a liquid above 1064 ☌). For this game, the most common isotopes of the chemical elements are used. This game presents a simple introduction to the Rutherford-Bohr model of the atom and the way we organize the elements. NS 5.8.2 - Develop an understanding of properties of matter Note: Any bond or intermolecular attraction that can be formed can be broken.Chemical and physical properties of materials can be explained by the structure and arrangement of atoms, ions, and molecules and the forces between them.Matter is made from discrete fundamental units called atoms. 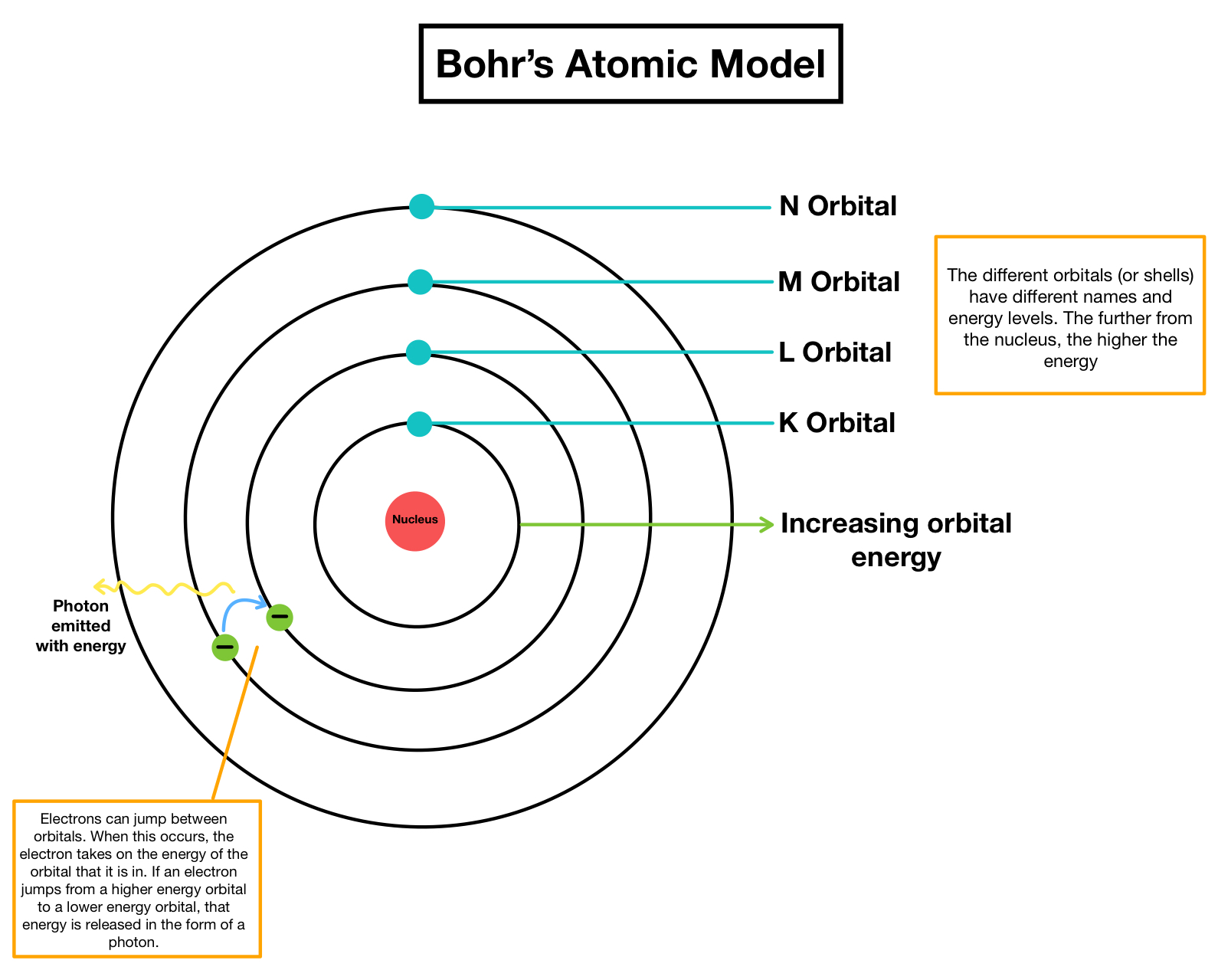
The existence of the atomic spectra is support for Bohrs model of the atom. Concepts:Ĭhemical elements, Periodic Table, chemical symbols, atom, atomic number, atomic weight, protons, electrons, neutrons, atomic structure. Bohrs model suggests that the atomic spectra of atoms is produced by electrons gaining energy from some source, jumping up to a higher energy level, then immediately dropping back to a lower energy level and emitting the energy difference between the two energy levels. Interactive module that introduces atomic structure Learning objectives:Īfter this activity, the student will be able to describe the basic structure of matter, name the parts of an atom, have experience using the Periodic Table, explain elements, and have the background to understand isotopes. A model of an atom according to Niels Bohr. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.Teacher Information: Activity Description: By Tereza Pultarova last updated 31 October 2022 The Bohr model is neat, but imperfect, depiction of atom structure. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. 
In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Electrons revolve around the nucleus in a. He concluded that electron will have more energy if it is located away from the nucleus whereas the electrons will have less energy if it located near the nucleus. The electrostatic force attracting the electron to the proton depends only on the distance between the two particles. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. According to Bohr Atomic model, a small positively charged nucleus is surrounded by revolving negatively charged electrons in fixed orbits. The simplest atom is hydrogen, consisting of a single proton as the nucleus about which a single electron moves. In atomic physics, the Bohr model if the atom (also known as the Rutherford-Bohr model) is modern model of the hydrogen atom introduced by Danish physicist Niels Bohr working with Ernest Rutherford at the University of Manchester in 1913. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
